Keita Ito and Willem Mulder receive ERC grant
The European Union will invest 5.25 million via the ERC grants for groundbreaking research into scoliosis and transplant organ rejection at the TU/e. The two projects will be led by Keita Ito and Willem Mulder, prominent researchers in the field of biomedical engineering.
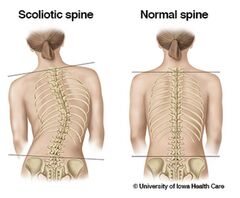
Advanced ERC grants are aimed at well-established top researchers, who have a recent high-level research track-record and profile which identifies them as leaders in their respective field. “With this generous award from the EU, we hopefully can provide a solution for this cruel disease, which causes disfiguration, mostly in otherwise healthy girls, during a crucial stage in their lives”, says Ito. Scoliosis (or adolescent idiopathic scoliosis (AIS), as its most common form is known) is a deformity of the spine that affects 2 to 3 per cent of the population. The disease mainly affects girls, who undergo earlier and more rapid growth than boys. It substantially reduces their quality of life and creates a life-long burden of disease. Although AIS has been known since the time of the ancient Greeks and despite centuries of dedicated research, no distinct cause has been identified.
One or multiple causes
“Some diseases have a single major cause, like Covid-19 or cystic fibrosis. However, many diseases are a product of many factors”, explains professor Keita Ito of the Department of Biomedical Engineering and leader of the research project. “We think that scoliosis or spine deformity in adolescence is such a disease, where a perfect storm brews during the rapid early growth spurt in girls.”
Together with colleagues of University Medical Center Utrecht, Ito hopes to create a paradigm shift in the field of scoliosis research,that will not only uncover the complex anatomical, biomechanical and mechanobiological causes of AIS, but also identify predictive triggersthat can be used for prevention and early treatment.
The research will combine bioengineering analysis (including imaging, in silico modeling, in vitro and ex vivo approaches in humans), and clinical medicine. One of the outcomes of the research will be a safe non-radiographic method to image the spine, which can become the new standard to monitor osseous injury and disease in juveniles in most hospitals.
Organ transplant acceptance
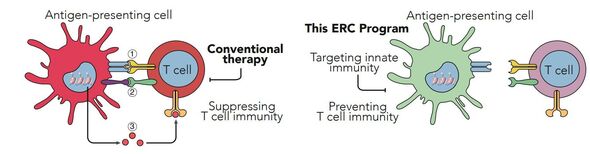
Together with his colleagues at RadboudUMC, Willem Mulder of the Department of Biomedical Engineering will explore novel nanotechnology to prevent organ rejection in patients who have received a transplanted organ. Organ rejection is caused by our immune system, which fights any foreign element that is introduced into our body.
While there are effective drugs on the market to suppress this reaction, chronic immunosuppression comes at a tremendous price. The drugs cause serious adverse effects, including cancer, and long-term rejection remains a serious problem. Rejection is mediated by innate immune system activation, which together with the adaptive immune system is responsible for our body’s defense against infections and other diseases.
Memory function
Recently, it has become clear that this innate system, just as our adaptive system, harbors a memory function, opening new avenues for treating illnesses like cancer. It is this innate system that Mulder and his colleagues also want to harness to increase our body’s tolerance to transplanted organs.
“The current strategy in organ transplantation is to suppress an already activated immune system, seriously compromising a patient’s ability to fight off disease. Our focus is on preventing immune system activation using highly innovative nanotechnology that integrates RNA or immunoregulatory proteins”, says Mulder.
While focusing on kidney transplant patients, the research can have wide-ranging benefits for other patients. “Similar innate immunity-focused disease management strategies can be developed for other conditions that are characterized by an exacerbated immune response. Think of COVID-19, autoimmune and cardiovascular diseases.”




Discussion