
Looking for life in Eindhoven’s primordial soup
A glass bowl gives us a glimpse of the future as well as of the past. Indeed, by trying to create life in that bowl, we may learn something about the original genesis of life. And you cannot consider that search for the origin of life separately from the question what life actually is - and what the future of life has in store for us. These grand issues are connected to a small – but long-term - experiment conducted by University Professor Bert Meijer and science museum NEMO.
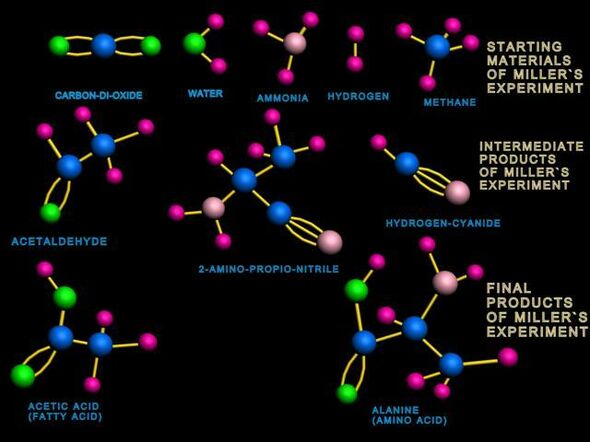
For five years a glass bowl, filled with water, methane, ammonia and hydrogen was staged in a display case in the Amsterdam science museum NEMO. During all that time, in an attempt to simulate lightning, there were sparks flying between two electrodes, which emerged from the glass like two devil’s horns. Three and a half billion years ago those outbursts of electric energy may well have been the driving force in the birth of the first organic molecules – the building blocks of life on earth as we know it.
The glass bowl formed a public experiment, an illustration of the type of experiment which was made famous in the early 1950s by pioneers Stanley Miller (PhD candidate at the time) and his supervisor Harold Urey (who had in 1934 been awarded a Nobel Prize for the discovery of deuterium). In the experiment carried out by Miller and Urey five different amino acids were discovered after a week – the building blocks of proteins. Later analyses, some of which were carried out only after Miller’s death in 2007, showed that at least 25 different amino acids had been formed. An impressive number, as there are only twenty occurring in nature.
As early as in 2009, on the initiative of Rob van Hattum, scientific director of NEMO, and Bert Meijer, Professor of Organic Chemistry at TU/e, a first version of the experiment was started. Supplementary to the experiment in NEMO, where the primeval soup would according to the planning remain untouched for five years, several experiments were planned at TU/e under varying circumstances. As something soon went wrong with the flask in NEMO – its neck was broken – and the experiments at TU/e proved to involve too much work after all, according to Meijer, a new attempt was made in 2012 - only in NEMO this time.
It goes without saying that as a public experiment the intention was for the primeval soup in NEMO to stay there longer than the week of Miller and Urey. Also, a decision was made for milder circumstances, says Bert Meijer in NEMO. “You have no idea what kind of requirements need to be satisfied by such exhibitions for children. Still, Miller and Urey heated their soup to nearly one hundred degrees. If you consider that the chemical reaction rate is almost doubled for every ten degrees the temperature is raised, and you know that the arrangement here is switched off during the evening and the night, then our five years resemble the week of the original experiment quite closely.”
Primordial soup under the microscope
In the spring of 2017 the arrangement in NEMO was dismantled and taken back to Eindhoven, where the contents were examined for the presence of amino acids.
The analysis of the contents - the ingredients – of the NEMO primeval soup was attributed to two Master students of Chemical Engineering and Chemistry; Marle Vleugels and Martin van Son. They did this in the form of a student assistant regime. Although this was an important assignment, there was no circle of scientists around them to see if they were doing thigs correctly when they opened the bowl after five years, says Van Son. “Marle and I had enough experience in the lab, also as student assistants in Bert Meijer’s group. Of course we worked in the most sterile way possible. First we unscrewed the electrodes and then we drew out the liquid from the bowl with a long, hollow needle. That liquid we preserved under a modified atmosphere in a number of small jars.”
In order to be able to give a definite answer about the formation, if any, of building blocks of life in the primeval soup, Vleugels and Van Son had to deploy a whole series of measurement techniques for the samples they had taken.
In addition to the liquid, which possibly contained amino acids, Vleugels and Van Son examined the deposit that had formed on the electrodes, as well as the substance that had been deposited in the liquid – originally pure water, which had turned brownish and turbid after five years. “We did not examine the gas, because all the interesting substances should be in the water, although we were also curious to find out everything about the solid matter: the deposit and the sediment.”
X-ray photoelectron spectroscopy
The students examined the solid matter by means of XPS (X-ray photoelectron spectroscopy). In this technique the matter is subjected to X rays and the electrons released in that process are observed. “We have looked at the elements carbon, nitrogen, oxygen and tungsten - the material the electrodes were made of”, Van Son explains. “We could see that the electrodes had oxidized, and that organic substances had formed - perhaps amino acids
Fourier-transform Infraroodspectroscopy
An analysis involving Fourier-transform Infrared spectroscopy (FTIR) showed that there were several substances in the sediment, which probably included silica from the glass. Which makes sense, because ammonia can slowly dissolve glass.”
Nuclear Magnetic Resonance
An NMR-spectrum (Nuclear Magnetic Resonance) subsequently showed that almost certainly several organic molecules had formed, which most probably included the amino acid glycine - the amino acid which Miller and Urey too had already found in the greatest quantities. Van Son: “We did see many peaks in the spectrum, but if you don’t know what exactly you are looking for, then NMR is not the most suitable technique.”
Mass spectroscopy
For this reason the students decided to unleash yet another measuring method on the primeval soup: mass spectroscopy. In this technique ionized molecules are accelerated by means of an electric field towards a detector. If that field is too weak, the trajectory of the molecules deflects too far down due to gravity, so that they miss the detector. As a result, the strength of the magnetic field whereby they hit the detector is a measure for the mass of the molecules - and indirectly for the composition.
Chromatography
Before you can begin with the mass spectroscopy, though, you first need to try and separate the molecules that you want to examine. “You do so by means of chromatography, whereby you send the molecules in a solvent through a column containing silica”, Van Son explains. Every molecule moves in its own tempo, so that they are each in their own place at the end of the column.
Derivatization
However, that too required an extra step: the so-called derivatization of the molecules. “Amino acids are not easy to detect with UV light, for instance, and we did want to do that. For that purpose we added a substance that lights up under UV light, and that binds to the amine group which contains each amino acid. That way we selected the possible amino acids at once as well.”
The molecules thus separated showed their identities in the mass spectrometer: in NEMO the amino acids glycine, alfa alanine and beta alanine had indeed been formed, just like in the original experiment. “Apart from those we have also found a small amount of 4-amino butyric acid, an amino acid that does not occur in nature and is even a bit more complex than those other three amino acids!”
New soup in triplicate
The NEMO experiment has been a success, then; apparently amino acids can also arise under very mild circumstances. And there are indications that possibly even more biologically relevant molecules have been formed, such as substances occurring in cell membranes. A graduating student will set to work with that now, according to Bert Meijer. Before those results may possibly be published, the experiment had to be repeated as far as he was concerned, “as ought to be done with every scientific experiment”.




Discussion