A miniature brain
The workings of our brain are still a mystery, to a great extent. Three-dimensional pieces of brain tissue on a chip could make research into brain diseases such as Alzheimer’s, Parkinson’s and epilepsy much simpler. That is why micro technologist Regina Luttge is working on special microbioreactors for brain cells, as platforms for a ‘brain-on-a-chip’. Recently she has been awarded a European Proof of Concept Grant in order to elaborate one of her findings further.
That the European Research Council (ERC) has awarded her a Proof of Concept Grant amounting to 150,000 euros, Regina Luttge, Associate Professor in the Microsystems (Mechanical Engineering) group, regards first and foremost as a recognition of the results she has achieved in the past four years. A Starting Grant which she received in 2011 from the same European science funder has enabled her to search for ways of culturing pieces of brain tissue in an artificial environment (in vitro) in three dimensions. That would signify a breakthrough, she explains.
“Neuroscientists working with in vitro models at present still have to make do with cell cultures in petri dishes. Those are actually two-dimensional systems, with a thickness of at most a few cell layers.” They cannot grow any higher, as the bottommost cell layers remain devoid of the nutrition that is added at the top of the dish. Real brains in fact have a complex three-dimensional structure; which is precisely why a flat section of brain tissue in a dish can hardly be said to be an optimal model system of our brain.
Luttge, who has vast experience in making so-called microfluidic systems – in which fluids are led through microscopic channels-, realized that these channels are about the same size as the capillaries through which oxygen and nutrients are transported to the cells in our organs. She decided to try and construct a kind of microfluidic platform in which cells can grow into genuine three-dimensional pieces of tissue.
She designed a cylindrical microbioreactor, with a diameter and height of several millimeters, in which the cells are fed via a microchannel along the circumference of the cylinder. Via a special membrane the channel is connected with a gelatinous substance inside the cylinder - the so-called scaffold -, in which the brain cells can grow. In this way even the innermost cells, located in the center of the cylinder, are still close enough to the nutrition to survive; in a cylinder with a greater diameter you would also need to create microchannels in the scaffold itself.

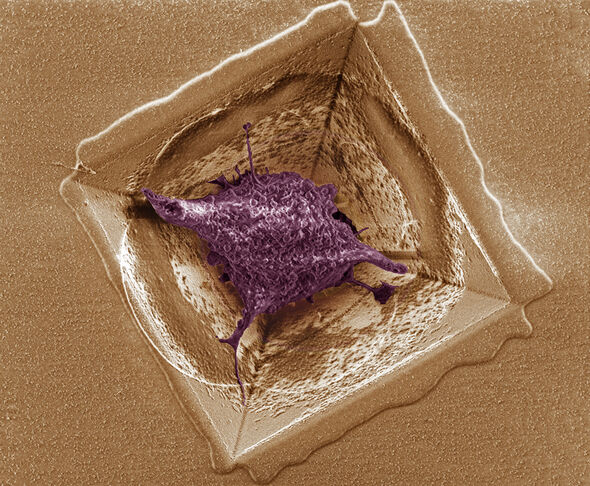
In order to be able to measure the electric activity of the brain cells, Luttge and her team manufactured a silicon plate provided with minuscule pores, evenly distributed across the surface. “The pores are in the form of an inverted pyramid”, the German scientist explains. “They form a kind of sieve with pits that have exactly the right size to catch a single brain cell per pit.” The brain cells nestle in the pits and feel very comfortable there, it appears.
“The cells in our pits stay alive for more than a week; they retain their natural, three-dimensional shape and develop protuberances by means of which they try to make contact with cells in neighboring pits.” By putting electrodes into the pits it becomes possible to measure the electric activity of these ‘healthy’ cells, says Luttge. “Moreover, by sending electric signals to the cells, it becomes possible in principle to disturb the system in a quite controlled manner, and subsequently you can see how the tissue reacts to that.”


Discussion