Home Stretch | Reaching the top of the ladder
Sometimes, chemistry is simply a matter of breaking apart molecules. In theory, this can be achieved by letting them vibrate until they break. Doctoral candidate Qin Ong demonstrated that you can convert CO2 to CO by irradiating CO2 with an infrared laser, a mechanism known as ‘ladder climbing’. This might even make it possible in the future to produce clean fuels.
Everyone who’s ever heard of the greenhouse effect will know that the excess concentration of CO2 in our atmosphere poses a huge problem. This is why it’s important to reduce the use of fossil fuels as fast as possible. Liquid fuels, however, have their advantages, and so far we haven’t managed to come up with a sustainable alternative for every application.
For this reason, it would be great if we could find a relatively simple way to convert CO2 into so-called solar fuels with the use of sustainably produced energy, such as solar power – so that CO2 will become part of a closed loop. The first step in that process is removing one of the two oxygen atoms (O) from the CO2 molecule. Because by combining the resulting CO with hydrogen, it becomes possible to produce combustible hydrocarbons.
Smart trick
However, CO2 is a highly stable molecule, Qin Ong explains. “You need temperatures of several thousands of degrees if you want to decompose it. This is extremely energy-intensive.” That is why scientists around the world are looking for a smart trick to split CO2 in an energy-efficient way. In Eindhoven, this is being done in TU/e’s Elementary Processes in Gas Discharges (EPG) group, and at energy institute DIFFER on the TU/e campus, where Ong carried out her doctoral research.
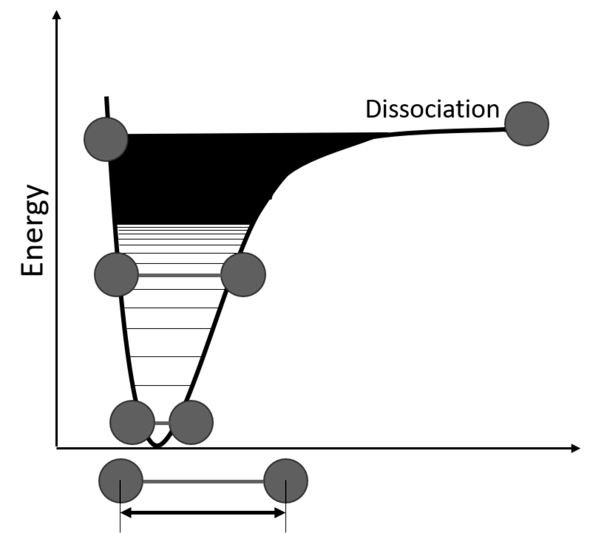
In one possible trick, scientists make use of the fact that atoms in a CO2 molecule can vibrate relative to each other, as if they’re connected to one another with a spring. “These vibrations only take place at specific frequencies,” Ong says. Each type of vibration has energy levels that you could think of as rungs of a ladder. The higher you climb the ladder, the harder the molecule starts to vibrate, and the less energy it takes to dissociate an atom from the molecule. They vibrate until they break, as it were.
Soviet experiments
Researchers at the EPG group – where Ong carried out experiments at the start of her doctoral period – are trying to make plasmas in which CO2 molecules climb the vibrational ladder by colliding with each other. “There are indications from old Soviet experiments that this can be a very efficient process for splitting CO2,”she says. “Originally, I was supposed to work on plasmas at DIFFER too, but we thought that we would be able to study the process of ladder climbing more accurately by stimulating the vibrations with laser light in normal CO2 gas. This happens to be a much simpler system than the one that requires you to use plasma.”
Infrared light allows you to push a CO2 molecule to the bottom rung of the vibrational ladder. Each time two molecules collide, one ascends a rung of the ladder, while the other one returns to the so-called ground-state. These steps follow on from each other at high speed, until the molecule breaks into pieces at the final collusion. “We know that this works well with molecules composed of two atoms,” Ong explains. “But with triatomic molecules such as CO2, it remained unclear whether the molecules would lose their energy during the process. Because if that were the case, they could never make it to the top of the ladder.”
Midnight
In order to test that, Ong – who was born in Nijmegen – needed a special laser from Radboud University named FELIX, which happens to be located at the department where she carried out her graduation research. “The FELIX laser allowed us to make light with the proper wavelength and intensity for my experiments.”
She couldn’t just drop by to take her measurements, by the way. “You need to submit a research proposal in order to get so-called beamtime from FELIX. I then spent a month preparing everything on location. Nothing can go wrong, because you can’t just come back the next day and do it all over again. They eventually gave us four nights to do our measurements. The laser operator leaves at midnight. If you’re lucky and the laser continues to operate properly, you get to continue with your measurement for a few more hours ‘for free.’ We did so until, two, three in the morning.”
Hopeful
The results of her measurements in Nijmegen, supplemented with measurements made with a laser at DIFFER, leave no room for doubt: the ladder climbing effect in CO2 does indeed exist. That’s hopeful, in part because the experiments were carried out at high pressure – of up to no less than 20 bar – and at a low temperature. “In our branch of chemistry, which deals with molecular vibration, collisions are usually thought of as a negative thing, which is why working at low pressure is more common. Because a collision between molecules can also cause them to lose their vibrational energy and fall from the ladder. According to my research, ladder climbing beats energy loss, even in the case of many collisions in experiments carried out at high pressure. That’s a very interesting result for this field.”
However, it remains to be seen whether Ong’s research will offer a solution for the production of solar fuels. “The aim was energy efficiency, and lasers use a lot of energy, unfortunately.” But who knows what the future will bring. “Researchers are working on LED lamps that emit the right infrared light. If they are energy efficient and intense enough, I see possibilities for the ladder climbing method.”



Discussion