Prof Talk | Vaccines against corona; how were they developed so fast?
Within a year after the pandemic broke out, people all around the world are already receiving vaccinations against COVID-19. How is this possible in such a short time? And doesn’t this compromise safety? Professor Willem Mulder and assistant professor Roy van der Meel of the Precision Medicine group at TU/e’s department of Biomedical Engineering know all the ins and outs. Spoiler: new techniques have accelerated the development of vaccines. “It’s logical that people have questions about the vaccines, but I wouldn’t hesitate for a moment to inoculate my family with it.”
Professor Willem Mulder perfectly understands that a significant number of people still aren’t that excited about getting vaccinated. “If you know that you usually have to wait ten years for a new vaccine, a healthy dose of mistrust is only natural. However, I’m not worried about safety issues, especially not with the mRNA nanovaccines developed by Pfizer/BioNTech and Moderna, which were the first to be approved by the European Union. It’s our task to explain how this new technology works and why it doesn’t pose any long-term safety risks.”
The Precision Medicine research group headed by Mulder and his colleague Van der Meel focuses on, among other things, new types of therapy based on nanoparticles and RNA. They also work at the iVAC initiative - within the Institute for Complex Molecular Systems (ICMS) - on a new platform aimed at developing new, nanotechnology-based vaccines (even) faster.
In short, they know what they are talking about. In fact: before he came to TU/e, in 2019, Van der Meel spent a few years working in Pieter Cullis’s group at the University of British Columbia in Vancouver, Canada. “That’s where the lipid nanoparticles were developed, best described as tiny fat globules, which are now used in the approved Pfizer/BioNTech and Moderna vaccines, as well as in an mRNA vaccine by CureVac that’s still underway,” the assistant professor says. “Those fat globules are one of the essential ingredients that make the mRNA vaccines such a success.”
Investment
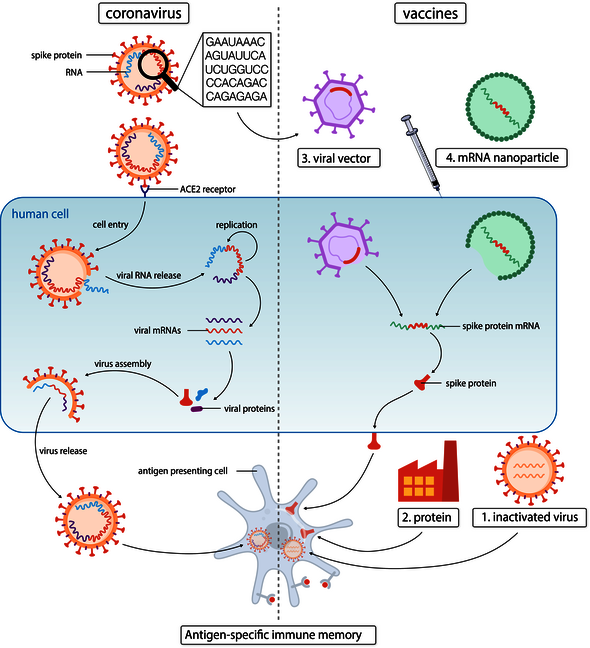
The sudden proliferation of corona vaccines is only partly the result of the massive investment of funding and resources that were made available immediately after the outbreak of the pandemic. Of equal importance is the fact that a number of vaccines based on new technologies were simply ready and eager to prove themselves; apart from the mRNA vaccines that hadn’t been applied before, these include the innovative vector vaccines (such as the Oxford vaccine developed by AstraZeneca) and protein vaccines (see illustration).
The goal of vaccinating against the coronavirus (top left in the illustration), it to teach immune cells (below) to recognize the virus, so that it will be rendered ineffective immediately after infection occurs. Traditionally, this is done with a weakened or inactivated (‘dead’) variant of the virus (1). Modern corona vaccines are based on the recognition of the so-called spike protein on the coronavirus’s surface. So-called protein vaccines contain spike proteins that are artificially produced based on the genetic code (in the form of RNA) from the virus itself (2). With vector vaccines, the related DNA instructions for the spike protein are inserted in the body cells via an innocent virus (the ‘vector’), after which the cell - via endogenous mRNA - starts to produce the spike protein on its own (3). With mRNA vaccines, the DNA step is skipped, and mRNA is injected artificially, locked in tiny fat globules (4).
The fact that we are no longer dependent on a weakened or inactivated (‘dead’) variant of the coronavirus thanks to these new vaccine development techniques, makes a significant difference. Van der Meel: “The virus needs to be sufficiently weakened, because vulnerable people might otherwise still get sick. And with a weakened or dead virus, you often need an ingredient that helps strengthen the immune response, a so-called adjuvant. Otherwise, the vaccine won’t work either. Finding out how far the virus should be weakened and finding the right adjuvants is often very time-consuming.”
An important reason why the current vaccines against the new coronavirus - officially SARS-CoV-2 - were developed at such a rapid pace, is because scientists had learned from earlier outbreaks of similar viruses SARS-CoV-1 (2003) and MERS (2012) that our immune system is able to recognize such corona viruses by the so-called ‘spike proteins’ at the virus’s surface. These protrusions, incidentally, are what give these viruses their name, because they resemble the protrusions on a crown (Latin: corona). SARS-CoV-2 has slightly different spike proteins, but the genetic code for this, contained in the virus’s RNA, can be quickly read with the use of modern sequencing techniques.
Chimpanzee virus
The Oxford vaccine developed by AstraZeneca is a vector vaccine, which uses an innocent - non replicating - adenovirus as a vehicle for DNA that encodes the spike protein. “They used a chimpanzee virus, not an adenovirus that already spreads among people,” Van der Meel emphasizes. “The advantage is that no one has antibodies against that adenovirus yet, because the vaccine will be much less effecitive if that were the case.”
That’s because our immune system would immediately render the adenovirus harmless, before it has a chance to instruct our body cells to produce the spike proteins. Incidentally, the makers of the Russian Sputnik vaccine, which uses two human adenoviruses, say that their vaccine doesn’t seem to suffer from that problem: they claim 95% efficacy. “In principle, these kinds of vaccines can be produced swiftly and cheaply,” Van der Meel explains. “Because it’s easy to grow such a vector virus, and it’s stable by nature, as a product of millions of years of evolution.”
Small protein factories
The mRNA nano vaccines from Pfizer/BioNTech and Moderna work much more directly. The instruction to produce the spike proteins - in the shape of artificial mRNA - is injected via tiny fat globules; there is no virus or DNA involved. And that turns out to yield remarkably good results.
This is quite wonderful, professor Mulder says: “No infection occurs, remember. There’s nothing that replicates and that might get out of hand due to an unexpected mutation.” Van der Meel adds: “The mRNA is read only a few times in the ribosomes, the small protein factories in the cell, whereby the decoded protein is produced each time. You have control over how many times a strand of mRNA can be read, by modifying the end of the mRNA molecule. After that, the process is completed and the mRNA is broken down. It stays in your body for no more than a few days.”
That transience is also the reason why Van der Meel isn’t worried about any possible long-term effects of mRNA vaccines either. The matter is settled, as far as he is concerned. “I want to get vaccinated as soon as possible. That includes my wife, and our newborn twins, should it be necessary.”
Mulder does however fear that immunity will last for a limited period of time after vaccination. “If you look at the course of the disease and the immunity induced by the infection itself, you have to wonder whether you’re protected for longer than a year.” In that case, an annual corona shot, not unlike the current flu shot for the elderly and the sick, might become a fact of life in the future.
Fat globules
If these mRNA vaccines work so well, why is it that they haven’t been commonplace for some time now? First of all, the necessary techniques to produce the mRNA haven’t been around for that long. Second, inserting the mRNA in the cell intact proved to be rather complicated. That puzzle has now been solved, in no small part thanks to Van der Meel’s former group in Vancouver.
“They came up with a technique to lock up the mRNA in fat globules that disintegrate when the acidity of their surrounding changes, such as in the body,” he explains. “As a result, it is now possible to produce large quantities of affordable mRNA vaccine. At the same time, the mRNA is released only inside the cell and it doesn’t trigger a dangerous immune response, but just the desired mild response.”
The first drug that delivered RNA contained in these kinds of fat globules to liver cells was approved in 2018, the assistant professor says. “The composition of mRNA vaccines against COVID-19 is based on this.”
I expect that Katalin Karikó and Drew Weissman will soon be awarded the Nobel Prize for their work on mRNA
Building blocks
However, once inside the cell, the vaccine-mRNA remains very vulnerable if it is taken for viral mRNA, because that will immediately be broken down. That is why certain building blocks in the artificial mRNA from both Pfizer/BioNTech and Moderna were replaced by a slightly different variant. As a result, the immune system inside our cells doesn’t even recognize it as mRNA (and therefore leaves it be), while it continues to very successfully play its role as a recipe for the production of proteins, surprisingly enough. “We have Katalin Karikó and Drew Weissman in particular to thank for those mRNA modifications. It is expected that they will win the Nobel prize for this within a few years.”
Should the current vaccines fail to protect against new strains of the coronavirus, such as the variants from South Africa and Brazil, the mRNA in the vaccine can be modified in an instant. “BioNTech's CEO has already said that they will be able to deliver the new vaccines within six weeks, should that be the case,” Van der Meel says.
In short: the development of the current types of vaccines based on new techniques was already quite advanced when the pandemic broke out, making it possible for the companies mentioned in this article to act swiftly. For example: the genetic code for the coronavirus was published online on 11 January 2020, and just one week later, Moderna and BioNTech were ready to test the first version of their vaccines.
Van der Meel: “The first trials on humans started in March already, and the pandemic suddenly freed up huge sums of money that made it possible to also start testing the efficacy and safety of vaccines on a large scale, the so-called phase 3.” Now that the technology has been proven, it can be expected that similar vaccines against other pathogens will be brought to the market more quickly - provided that the need is urgent enough.




Discussion